 
Phosphorus has three main allotropes: white, red and black. This is the material, mixed with powdered glass, stuck on the side of boxes of safety matches on which the matches must be struck to light them. Red phosphorus, made by gently heating white phosphorus in the absence of air to about 250☌, does not glow, is stable and is not poisonous. White phosphorus is manufactured industrially, glows in the dark, is spontaneously flammable when exposed to the air above 30☌ and is a deadly poison. Pure phosphorus is obtained by heating a mixture of phosphate rock, coke, and silica to about 1450 ☌. 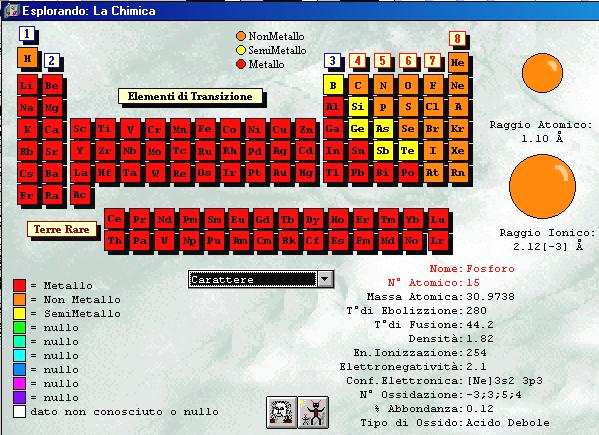
Elementary phosphorus is liberated as vapor and may be collected under phosphoric acid, an important compound in making super-phosphate fertilizers.įound most often in phosphate rock. By one process, tri-calcium phosphate, the essential ingredient of phosphate rock, is heated in the presence of carbon and silica in an electric furnace or fuel-fired furnace. White phosphorus may be made by several methods. The red modification is fairly stable, sublimes with a vapor pressure of 1 atm at 17C, and is used in the manufacture of safety matches, pyrotechnics, pesticides, incendiary shells, smoke bombs, tracer bullets, etc. It should, however, be handled with care as it does convert to the white form at some temperatures and it emits highly toxic fumes of the oxides of phosphorus when heated. This form does not ignite spontaneously and is not as dangerous as white phosphorus. When exposed to sunlight or when heated in its own vapor to 250oC, it is converted to the red variety, which does not phosphoresce in air as does the white variety. White phosphorus should be kept under water, as it is dangerously reactive in air, and it should be handled with forceps, as contact with the skin may cause severe burns. Exposure to white phosphorus should not exceed 0.1 mg/m3 (8-hour time-weighted average - 40-hour work week). It is very poisonous, 50 mg constituting an approximate fatal dose. It takes fire spontaneously in air, burning to the pentoxide. It is insoluble in water, but soluble in carbon disulfide. White phosphorus has two modifications: alpha and beta with a transition temperature at -3.8oC. Ordinary phosphorus is a waxy white solid when pure it is colorless and transparent. 
Phosphorus exists in four or more allotropic forms: white (or yellow), red, and black (or violet). P-red is largely amorphous inorganic polymer, although X-rays have established the existence of several crystalline forms, normally present in a limited extend (<10%w). The colour ranges from the orange to the dark violet depending on molecular weight, particle size and impurities. The term Red-phosphorus (P-red) is used for describing one of the allotropic forms of Phosphorus, obtained by heating White Phosphorus (P-w) at a temperature close to 300C in absence of oxygen. 
htm Very good info on physical properties. The reaction may proceed via "phosphorus pentoxide", P4O10. The usial route involves heating a phosphate with sand and carbon in an electric furnace. There is normally no need to make phosphorus in the laboratory as it is readily available commercially. However there is plenty of phosphorus in phosphate ores and those ores represent the usual source for commercially produced phosphorus. Originally, phosphorus was extracted from urine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
